– Impingement Cooling
– Impingement Jet Array Heat Transfer With Surface Textures
– Surface Roughness Characterization and Analysis
– Dimple Surface Arrays
– Swirl Chambers
– Surface Heat Transfer Augmentation Within Internal Passages
– Film Cooling
– Full-Coverage Film Cooling
– Double Wall Cooling
– Second Law Analysis of Film Cooling
– Aerodynamic Losses From Turbine Airfoils
– Transonic Turbine Blade Tips
– Transonic Turbine Blade Tips With Film Cooling
– Transonic Turbine Alloy Blades
– Supersonic Flow Experimental Results
– Shock Wave Unsteady Interactionss
– Viscous Dissipation Within a Transonic Flow Environment
– Transitional Flows in Curved Channels
– Dean Flow Dynamics in Low-Aspect Ratio Spiral Microchannels
– Unsteady Laminar Impinging Slot Jets
– Electronics Cooling
– Miniature and Micro-Scale Pumps
– Slip Rarefaction Phenomena
– Elastic Instabilities
– Buoyancy-Driven Continuous SPLITT Fractionation
BOUYANCY-DRIVEN CONTINUOUS SPLITT FRACTIONATION
Buoyancy-Driven Continuous SPLITT Fractionation
REFERENCE:
Buoyancy-Driven Continuous SPLITT Fractionation: A New Technique for Separation of Microspheres (J. Storey, P. Douglas, P. M. Ligrani, and K. M. Morten), Separation Science and Technology, Vol. 44, No. 9, pp. 1895-1922, January 2009. DOWNLOAD COPY OF PAPER
FIGURES:
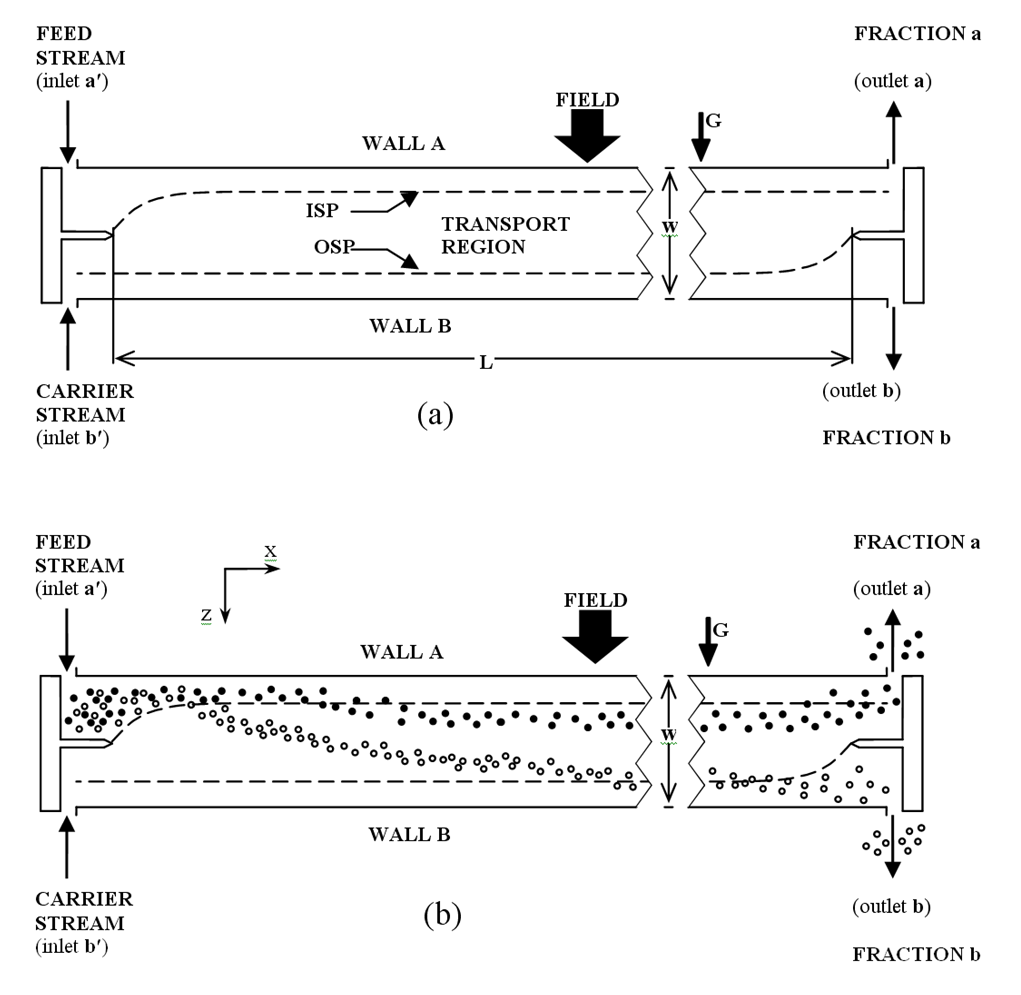 Schematic view of a SPLITT cell operating in sedimentation transport mode, showing (a) the overall layout of the cell, and (b) example particle trajectories during operation.
Schematic view of a SPLITT cell operating in sedimentation transport mode, showing (a) the overall layout of the cell, and (b) example particle trajectories during operation.
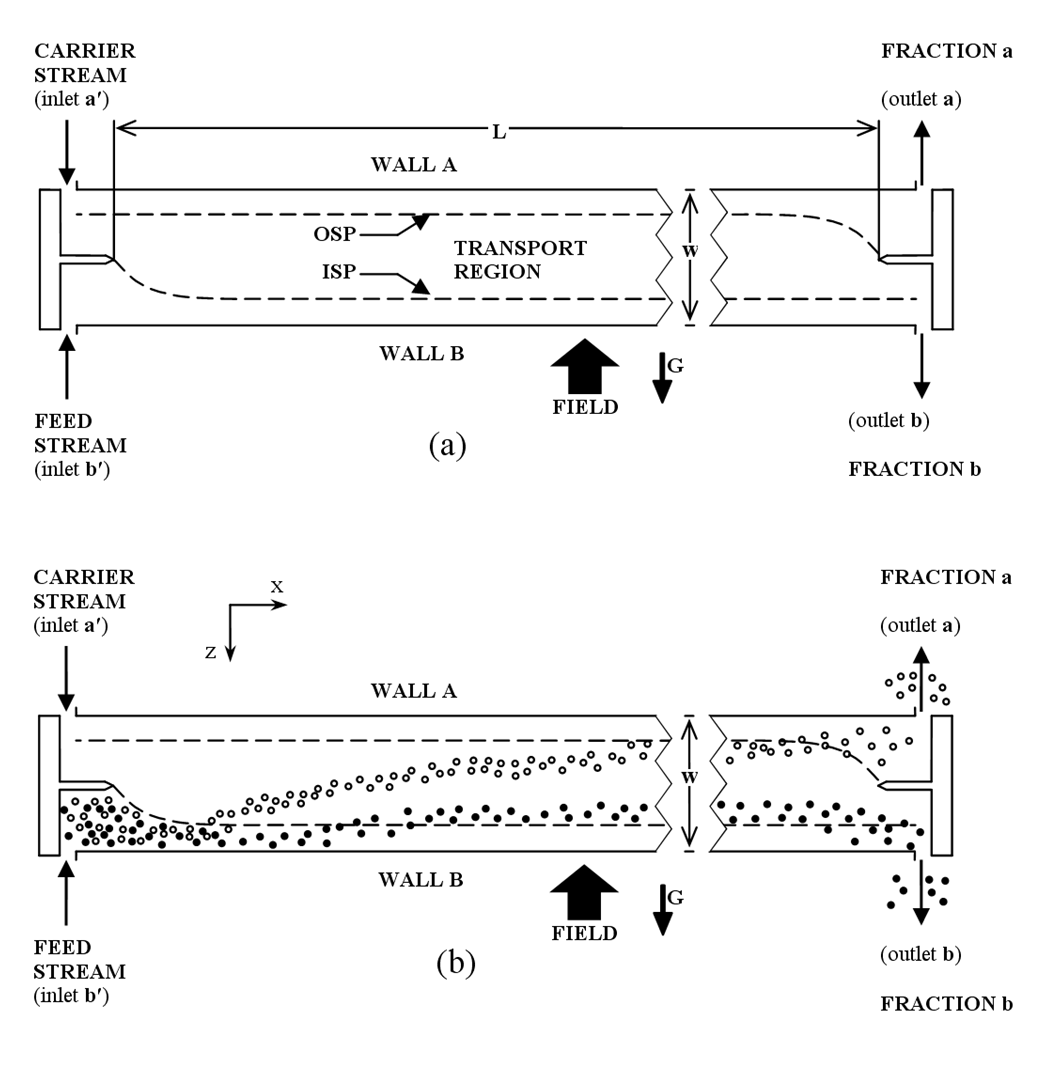 Schematic view of a SPLITT cell operating in buoyancy-driven transport mode, showing (a) the overall layout of the cell and (b) example particle trajectories during operation.
Schematic view of a SPLITT cell operating in buoyancy-driven transport mode, showing (a) the overall layout of the cell and (b) example particle trajectories during operation.
 Photograph of the cell for the sedimentation-driven and buoyancy-driven experiments.
Photograph of the cell for the sedimentation-driven and buoyancy-driven experiments.
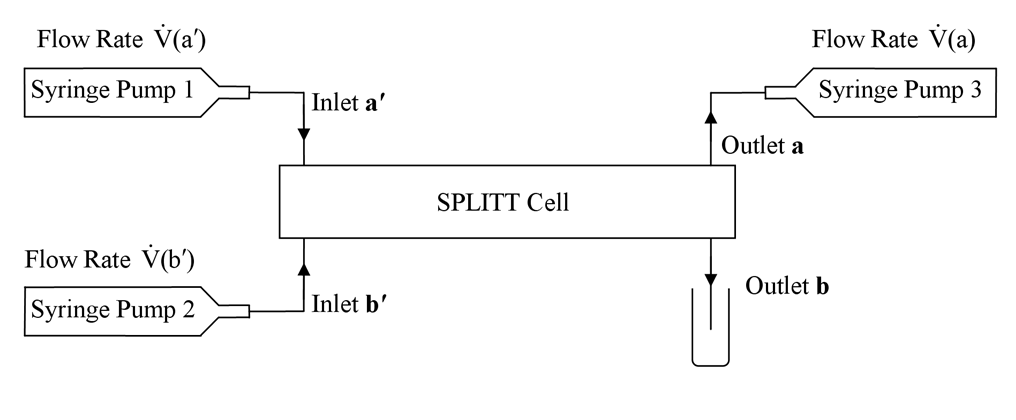
Schematic diagram showing flow connections to and from the cell, and the arrangement of the syringe pumps.
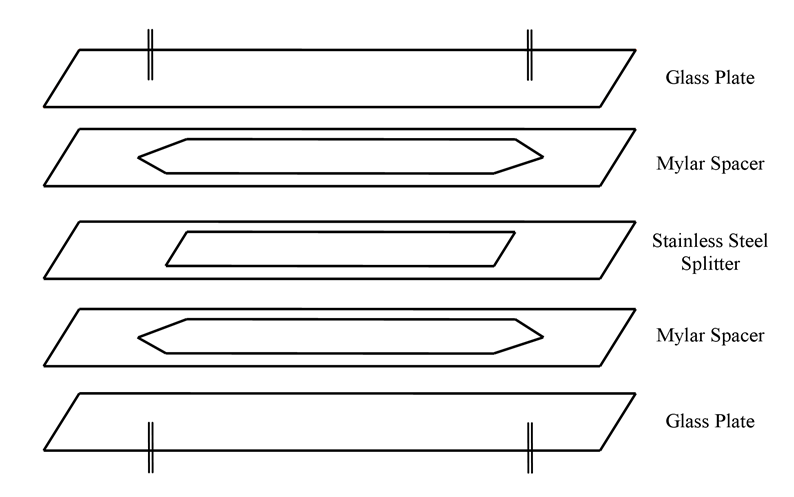 Schematic diagram showing an overview of the cell construction and layout.
Schematic diagram showing an overview of the cell construction and layout.
RELATED REFERENCES:
Resolution Deterioration and Optimal Operating Conditions in Centrifugal SPLITT Fractionation. Part I: Stable Density Gradients (S. Gupta, P. M. Ligrani, M. N. Myers, J. C. Giddings), Journal of Microcolumn Separations, Vol. 9, No. 3, pp. 213-223, March 1997. (Errata: Vol. 9, No. 6, p.521, June 1997.)
Resolution Deterioration and Optimal Operating Conditions in Centrifugal SPLITT Fractionation. Part II: Unstable Density Gradients (S. Gupta, P. M. Ligrani, M. N. Myers, J. C. Giddings), Journal of Microcolumn Separations, Vol. 9, No. 4, pp. 307-319, April 1997. (Errata: Vol. 9, No. 6, p.521, June 1997.)
Investigations of Performance Characteristics Including Limitations Due to Flow Instabilities in Continuous SPLITT Fractionation (S. Gupta, P. M. Ligrani, and J. C. Giddings), Separation Science and Technology, Vol. 32, No. 10, pp. 1629-1655, October 1997.
Onset and Effects of Instabilities From Unstable Stratification of Density on Mass Transfer in Channel Shear Layers at Low Reynolds Numbers (P. M. Ligrani, S. Gupta, and J. C. Giddings), International Journal of Heat and Mass Transfer, Vol. 41, No. 12, pp. 1667-1679, June 1998.
Characteristics of Flow Instabilities From Unstable Stratification of Density in Channel Shear Layers at Low Reynolds Numbers (S. Gupta, P. M. Ligrani, and J. C. Giddings), International Journal of Heat and Mass Transfer, Vol. 42, No. 6, pp. 1023-1036, March 1999.
